|
The anode attracts anions and reduces them. In Galvanic cells and electrolytic cells, the cathode attracts cations and oxidises them. In Galvanic Cells and Electrolytic Cells: Whereas reduction takes place at cathodes. In devices that rely on redox reactions, oxidation takes place at the anodes. This means that outside the device, electrons flow from anode to cathode. Generally, current flows into the anode from the outside.Ĭathode gives current out of the device. 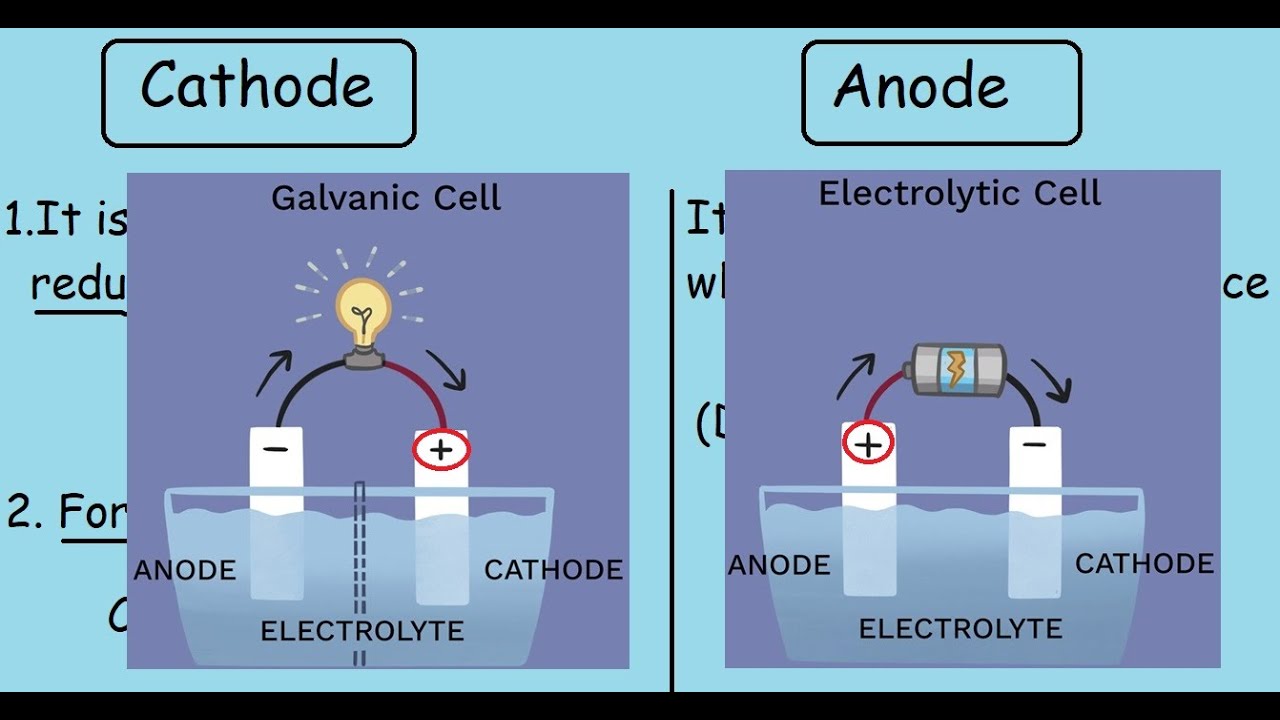 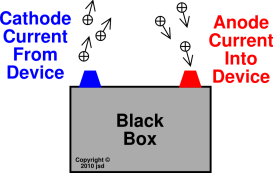
A few more examples are mentioned in the summary section below. We have discussed two particular examples from electrochemistry but the terms “anode” and “cathode” are used in many other fields as well. If possible, alternative, less ambiguous terms should be used (depending on the situation). Therefore, it may be necessary to first examine the situation in order to use the terminology properly. However, because the way currents flow in different situations could be radically different, translating the use of these terms from one situation to another could be confusing. The names “anode” and “cathode” can be given to a terminal depending on whether current flows into that terminal from outside, or whether current flows out of the terminal to the outside. For example, the “positive” electrode in the Galvanic cell is its “cathode”, but the “positive” electrode in the case of electrolysis is its “anode”. The above two examples should clarify that the terms anode and cathode do not refer to a specific potential, but rather how current flows in the setup. Therefore, this terminal forms the cathode. Current flows out of the device from this terminal. Sodium forms at the other electrode where ions are reduced. ions lose their electrons here, so this is consistent with the idea that oxidation happens at the anode. Here, the terminal that draws current into the device is the electrode connected to the positive terminal of the battery. Here, these ions give off their electrons, forming chlorine gas.Īt the electrode connected to the negative terminal, the positive sodium ions gain electrons, forming sodium atoms: The electrode connected to the positive terminal of the battery attracts the anions. For instance, we will look at what happens when two electrodes are put into a sample of molten sodium chloride (NaCl, or common salt). In electrolytic cells, a power supply is used to create a current in a liquid containing ions. This agrees with the description above: zinc (the anode) becomes oxidised and copper (the cathode) gets reduced. Whenever a device works using redox reactions, the terminal where oxidation happens is the anode, and the electrode where reduction happens is the cathode. Conventional current flows out of the device at the copper terminal, so that makes copper the cathode. Here, electrons flow “out of the device” from the zinc terminal, so the conventional current is flowing into the device here. Copper is being reduced (it gains electrons): Here, the incoming electrons combine with Cu 2+ ions and form copper atoms. The electrons lost by zinc flow across the wires onto the copper electrode. In other words, zinc is being oxidised (it loses electrons). At the zinc electrode, the zinc dissociates into Zn 2+ ions and electrons. In the cell drawn above, copper has a higher reduction potential than zinc, so it draws electrons from the zinc electrode. The electrode with a higher reduction potential has a stronger ability to gain electrons, so electrons flow into it from the other electrode. 
In a Galvanic cell, one of the electrodes is at a higher reduction potential than the other. This means that electrons flow out of the device at the anode. What is an AnodeĪnode is the terminal where the (conventional) current flows into a device from outside. In this article, we will look at several scenarios where these terms are used, and explore their usage in terms of the processes that occur in these devices. Undoubtedly, this may lead to confusion and it is advisable to adapt to the general usage in the specific field. However, the usage is not strictly followed in some instances, because when a device is able to undergo a reversible process, the same terminal that was called the “anode” could be now called the “cathode”. The terms cathode and anode are used to refer to terminals of a polarised electrical device. The main difference between anode and cathode is that, in general, anode is the terminal where the (conventional) current flows into a device from outside, whereas cathode is the terminal where (conventional) current flows out of the device.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
